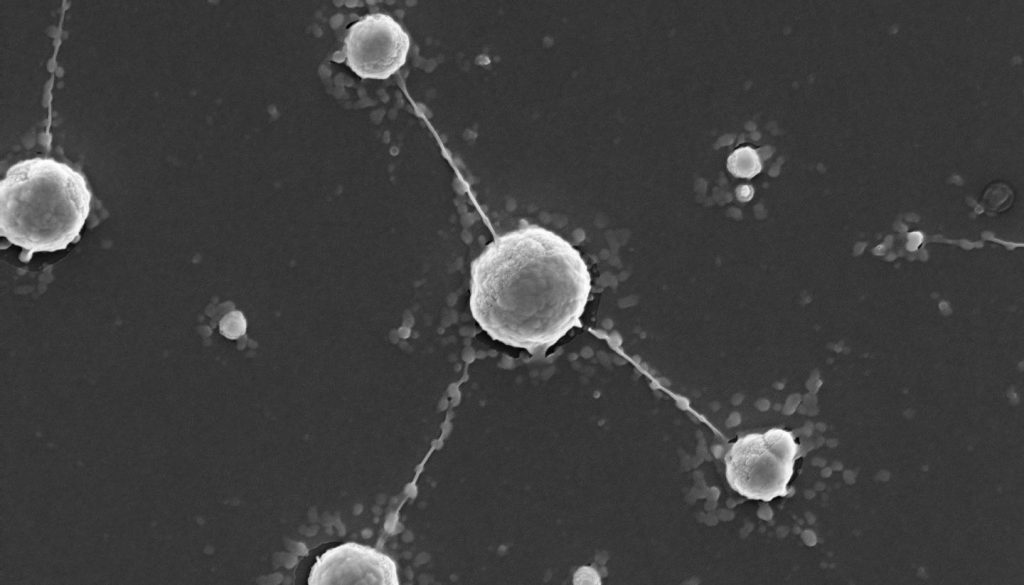
Do bacteria use tungsten for protection from interstellar radiation?
Tungsten is a heavy metal with impressive properties: the white, shiny material doesn’t melt until the temperature is at 3422 °C and doesn’t boil until 5930 °C. It is resistant to almost all acids and has approximately the density of gold. It is also interesting that all its natural isotopes are theoretically unstable. Their half-lives, however, are on the order of trillions of years, so their decomposition is not measurable on our time scales. Humans have used tungsten to construct light-bulb filaments for incandescent and fluorescent lights. In the carbon compound, tungsten carbide, it is almost as hard as…