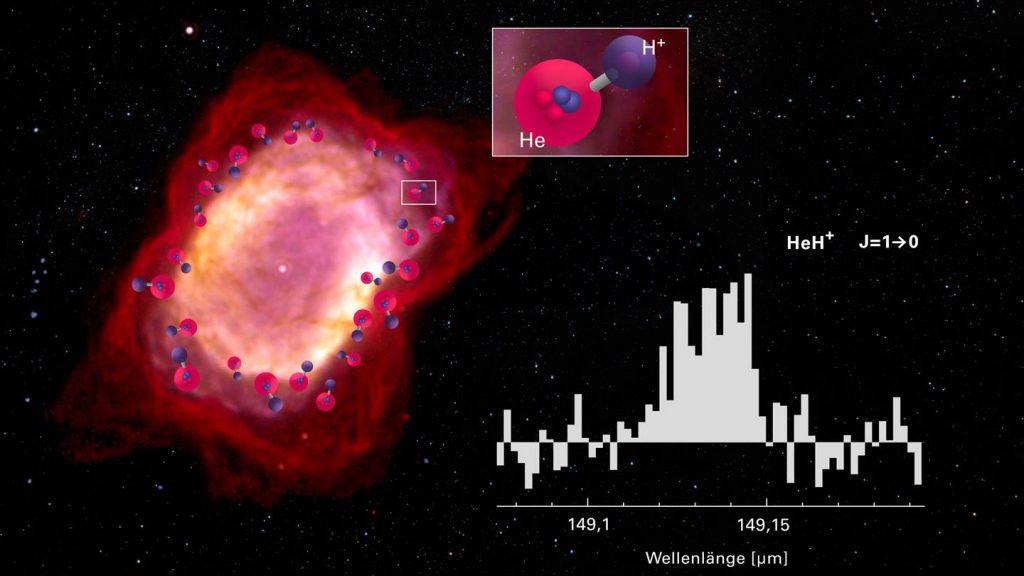
Airborne telescope detects helium hydride ion in space
The helium hydride ion HeH+ is a puzzle in and of itself. As a noble gas, helium does not easily bond with other elements. And in the early universe, the selection of elements was much smaller than it is today: the only elements were hydrogen (H), helium (He), and traces of lithium, and only in ionized form, that is, without electrons, which form the basis for chemical bonds. After the big bang, the universe had to cool down first, for a period of approximately 300,000 years, before chemistry could begin. At a temperature of about 3700 degrees Celsius, the…